Elevated Vigor. Visible Refinement.
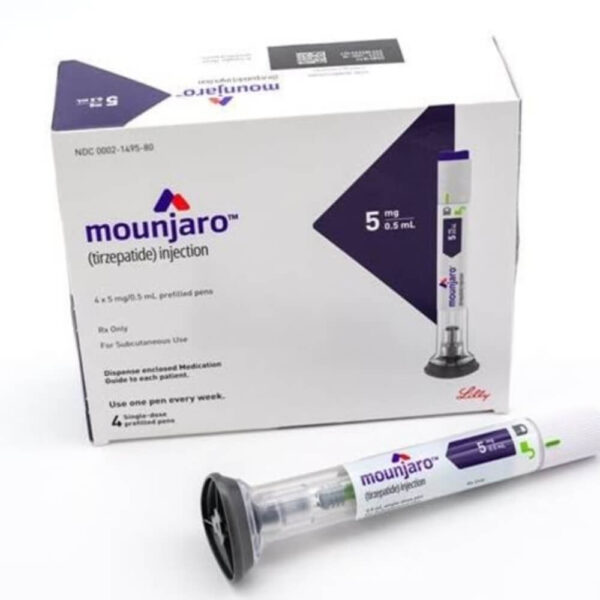
Mounjaro® (Tirzepatide) Injection — Once-Weekly Dual-Action Pen
Price range: $199.00 through $400.00
Extra Features
- Dermatologist Approved
- Secure Payments
- Satisfaction Guarantee
- Worldwide Shipping
- Money Back Guarantee
Type 2 diabetes · Dual GIP + GLP-1 receptor agonist · Subcutaneous autoinjector
Mounjaro® 2.5 mg — Where Dual-Action Therapy Begins
Once-weekly pen · 4-week initiation dose
Mounjaro® 5 mg— Double the Receptors, Real Measurable Results
Once-weekly pen · First maintenance dose
Mounjaro® 7.5 mg — Midcourse Dual-Incretin Escalation
Once-weekly pen · Intermediate escalation dose
Mounjaro® 10 mg — High-Gear Dual-Receptor Engagement
Once-weekly pen · Higher maintenance dose
Mounjaro® 12.5 mg— Near the Ceiling of Dual-Incretin Power
Once-weekly pen · Penultimate escalation dose
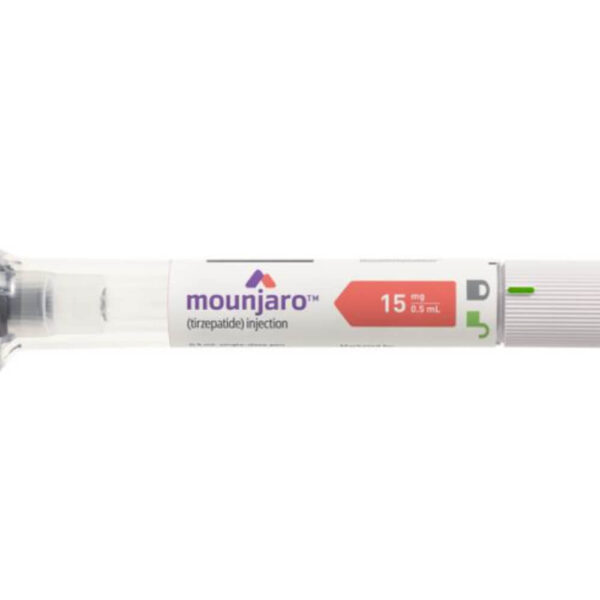
Mounjaro® 15 mg — Peak Dual Power, Once a Week
Once-weekly pen · Maximum approved dose
The starting point for tirzepatide’s twin-receptor approach — a gentle introduction to GIP and GLP-1 working together for the first time in your system.
Mounjaro® 2.5 mg tirzepatide is the four-week starter dose, initiating dual GIP and GLP-1 receptor agonism with minimal gastrointestinal side effects. Once-weekly subcutaneous injection via autoinjector establishes the base for the structured escalation protocol in type 2 diabetes management.
The first true maintenance milestone. Dual-receptor activity is now in full swing — blood sugar improvements become clinically measurable week over week.
Mounjaro® 5 mg tirzepatide is the initial maintenance dose for type 2 diabetes, delivering once-weekly dual incretin stimulation. This dose has demonstrated significant HbA1c reduction and a weight loss advantage over single-receptor GLP-1 therapies in the SURPASS clinical program.
The midpoint of Mounjaro’s dose ladder — where glycemic control deepens and weight loss often accelerates most noticeably for patients.
Mounjaro® 7.5 mg tirzepatide is an optional intermediate escalation dose for patients who benefit from gradual advancement toward higher therapeutic levels. Administered once weekly via pre-filled autoinjector pen as part of the standard titration schedule in type 2 diabetes management.
Powerful dual-receptor engagement at a high therapeutic level — proven to cut HbA1c by over two percentage points on average in clinical trials.
Mounjaro® 10 mg tirzepatide delivers high-intensity dual GIP and GLP-1 receptor agonism once weekly, demonstrating superior HbA1c reductions and body weight loss compared to approved comparators in the SURPASS clinical trial program.
Approaching maximum potency — one step before the highest approved dose for those who need the most from tirzepatide therapy.
Mounjaro® 12.5 mg tirzepatide offers near-maximum dual-receptor stimulation for intensified type 2 diabetes management. Once-weekly subcutaneous delivery continues preparing patients for the highest approved maintenance dose when additional glycemic control is required.
The highest -approved tirzepatide dose — the full force of two incretin pathways working in concert, one injection per week.
Mounjaro® 15 mg tirzepatide is the maximum approved dose, achieving the most profound dual GIP and GLP-1 receptor agonism available. In the SURPASS trials, this dose produced average HbA1c reductions of approximately 2.4% and body weight loss of up to 22.5 lbs, delivered via once-weekly subcutaneous autoinjector pen.
| Dosage | 2.5 mg, 5mg, 7.5mg, 10mg, 12.5mg, 15mg |
|---|



Reviews
There are no reviews yet.